Details
Engineered enzyme
Engineered reverse transcriptases allowed the synthesis of cDNA from very low amounts of RNA. Mutations are inserted into the RNase H domain of the MuLV‘s reverse transcriptase. Therefore, by not degrading the RNA during the first-strand synthesis, a higher yield of full-length cDNA is obtained. Additionally, a higher thermal stability increases the robustness of the enzyme. The FastGene® Scriptase II is exactly one of those engineered enzymes. With its mutation in the RNase H domain and higher thermal stability, it is the optimal choice for more complex applications, such as RT-qPCR and NGS.
Applications
- Quantification of Gene Expression
- RT-qPCR
- Next Generation Sequencing
- Low RNA concentration
- Difficult templates
Lower RNase H activity for longer cDNA
The FastGene® Scriptase II has a modified RNase H domain. The RNA is therefore not degraded and serves as a template for longer cDNAs, resulting in fragment size of up to 12 kBp.
Engineered enzymes – optimized for qPCR
The FastGene® Scriptase II delivers superior cDNA templates for downstream applications, e.g. qPCR and NGS. The resulting full-length cDNA gives a complete picture of the gene and is able to show modification, e.g. splicing variants.
Comparison of different PCR results
Multiplex PCR
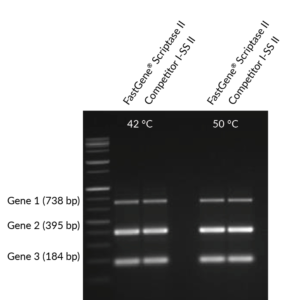
Fig. 1: Comparison of multiplex PCR using cDNA produced by Competitor I‘s SS-II enzyme and FastGene® Scriptase II at 42 °C and 50 °C.
qPCR
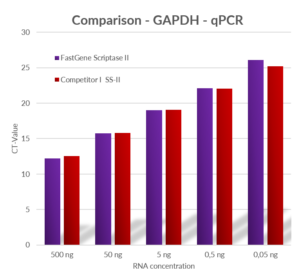
Fig. 2: Comparison of qPCR results using primers for GAPDH and cDNA produced by using different RNA starting concentration by Competitor I‘s SS-II enzyme and FastGene® Scriptase II at 42 °C.
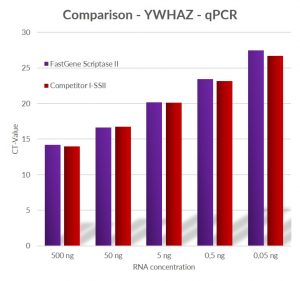
Fig. 3: Comparison of qPCR results using primers for YWHAZ and cDNA produced by using different RNA starting concentration by Competitor I‘s SS-II enzyme and FastGene® Scriptase II at 42 °C.





Haruko Hayasaka Immunohole Functional Lab., Dept. Bioscience and Biotechnology, Kinki University –
I especially like that the Scriptase II lead to stable results. As a result of performing RT-PCR using tumor derived RNA, we were able to detect the expression of genes whose amplification was unstable with other RT reagents. The Amplification of full-length cDNA has also been confirmed. I would love to also try the 5x Ready Mix.
Verified User
Ryo Mameda, Department of Biomolecular, Graduate School of Engineering, Tohoku University, Japan –
PCR amplified DNA fragement was cloned into a vector and the sequence was confirmed. As a result, there was no artificial mutation such as changes of single nucleotides.
Also, the manual of your product (FastGene® Scriptase II cDNA Synthesis Kit) was very easy to understand and I felt no stress on the operation. I definitely want to use other products from NIPPON Genetics.
Verified User
Dr. Catherine Moermans, pneumology department, CHU-ULiège, Liège, Belgium –
“The FastGene® scriptase II cdna synthesis kit appeared to be a reliable method to perform RT-PCR experiments using induced sputum samples which is a difficult specimen matrix. Indeed, it allowed to obtain a good reproducibility and good amplification curve profils. Furthermore, the cost of this product is really cheap compared to what we use to buy.”
Translation by Nippon Genetics:
Das FastGene® Scriptase II cDNA Kit zeichnet sich als zuverlässige Methode aus um RT-PCR Experimente mit Proben eines induzierten Suptums durchzuführen. Obwohl dies eine schwere Probenmatrix ist, konnte eine gute Reproduzierbarkeit und ein gutes Amplifikationskurvenprofil gewonnen werden. Zusätzlich sind die Kosten verglichen zu dem zuvor verwendetem Produkt sehr preisgünstig.
Verified User
Nathalie Renotte, Cellular and Molecular Epigenetics, GIGA Cancer, Liège, Belgium –
“We have tested the RT kit FastGene Scriptase II in comparison with other kits, like Protoscript II (NEB) and Superscipt II (Invitrogen) and we have noted that the results are similar. Additionally, we can make more “runs” for a better price!”
Verified User