Details
BlueStar Prestained Protein Marker
The BlueStar Prestained Protein Ladder is a three color protein standard with 10 prestained proteins covering a wide range molecular weights from 10 to 180 kDa. The BlueStar Prestained Protein Ladder is designed for monitoring protein separation during SDS-polyacrylamide gel electrophoresis, verification of Western transfer efficiency on membranes (PVDF, nylon or nitrocellulose) and for approximating the size of proteins.

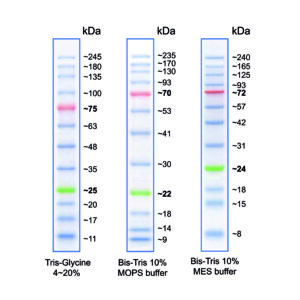
Figure 1: Molecular weight of the marker bands when using different buffer systems.
The molecular weight of the marker bands can vary depending on the gel/buffer system used. This is due to factors of the gel/buffer system that affect the running behaviour of proteins, e.g. migration rate, gel composition, pH-value (see FAQ for more information).
- Broad range: 10 – 180 kDa
- High performance: 10 bands with 3 colours (blue, orange and green)
- Reference bands: 28 kDa (green) and 75 kDa (orange)
- Sharp bands
- Ready to use: Supplied in a loading buffer for direct loading
Storage Buffer
20 mM Disodium hydrogen phosphate pH 8.0 (at 25°C ), 2 % SDS, 0.2% Triton X-100,10 mM 2-Mercaptoethanol, 6 M Urea and 27% (v/v) Glycerol.
Storage
Stable at -20°C for 12 months.
Stable at +4°C for 3 months.
Recommendations for Loading
1. Thaw the ladder either at room temperature or at 37-40°C for a few minutes to dissolve precipitated solids. Do not boil.
2. Mix thoroughly, to ensure that the solution is homogeneous.
3. Load the following volumes of the ladder on SDS-polyacrylamide gel:
- 5 μl per well for mini-gels, 3 μl per well for blots
- 10 μl per well for large gels, 6 μl per well for blots
Applications
• Monitoring of protein migration during SDS-polyacrylamide gel electrophoresis
• Monitoring of protein transfer onto membranes during Western blotting
• Sizing of proteins on SDS-polyacrylamide gels and Western blots
FAQ
Does this marker contain Formaldehyde or Formamide?
No.
Why are the molecular weights of the marker bands for the protein markers given separately for different systems?
There are several factors of gel and buffer chemistry that have an influence on the running behaviour of proteins. The most important are:
Migration rate: The migration of proteins in electrophoresis is influenced by the properties of the buffer system, e.g. by the ionic strength, the pH value and the presence of certain ions. The different buffer systems influence the electrophoretic mobility of proteins differently. For example, proteins may migrate faster or slower in one buffer system than in another, resulting in obvious differences in their molecular weight.
Gel composition: The type of polyacrylamide gel and its pore size can influence protein migration. Different buffer systems may be optimised for different gel types, which affects how proteins move through the gel matrix. These differences can lead to variations in the observed molecular weights.
pH: The pH of the electrophoresis buffer can also affect the charge and conformation of the proteins, which in turn affects their migration in the gel. Different buffer systems may have different pH values, leading to variations in molecular weight estimates.
Tris-glycine gels contain a moving front of Cl- ions (from the Tris-HCl in the gel) and negatively charged glycine ions from the running buffer. The glycine ions move slower than the chloride ions through the low pH (pH 6.8) collection gel and form a narrow zone where the proteins are concentrated. When the moving front meets the separating gel with the higher pH (pH 8.8), the glycine ions move quickly past the chloride ions, pulling the proteins through the gel with them, so to speak. Because of this composition, tris-glycine gels work in the strongly alkaline range and reach a pH of up to 9.5 when running.
Only logged in customers may leave a review.








Norbert Zanker, Pharmakologie und Toxikologie, Universität Ulm –
Very good product. The range of the protein ladder is for our purposes perfect.
Nicely visible protein ladder.
We use it as our standard protein ladder for gel electrophoresis.
Translation by Nippon Genetics:
Sehr gutes Produkt. Der Bereich der Protein Ladder ist für unsere Zwecke perfekt. Eine gut sichtbare Protein Ladder. Wir nutzen diesen Protein Marker als unseren Standard für die Gelelektrophorese.
Verified User
PhD Marina Cotsiki, Molecular Oncology Laboratory, BSRC Alexander Fleming, Greece –
We have been using Nippon Genetics protein markers in our laboratory for the past 7 years. They are easy to use and exceptionally cost-efficient. The range of the molecular weights is very broad so we can use them for all of our SDS-polyacrylamide gel applications. The bands are sharp and focused and very easily identifiable by the reference colors. Overall, we are extremely satisfied with the quality of these markers.
Translation by Nippon Genetics:
Wir benutzen die Protein Marker von Nippon Genetics seit 7 Jahren in unserem Labor. Sie sind leicht anzuwenden und besonders preisgünstig. Der Molekulargrößenbereich ist sehr umfassend, so dass wir die Marker für alle unsere SDS-Polyacrylamidgelapplikationen benutzen können. Die Banden sind scharf, klar und durch die Referenzfarbbanden sehr einfach identifizierbar. Insgesamt sind wir sehr zufrieden mit der Qualität dieser Marker.
Verified User